 The ionic radii increase down a group because there is an extra energy level for electrons as you travel down the group of elements.Ģ. Why do ionic radii increase down a group? When electrons are added together, they form an ion that is bigger than the source element.ġ.When electrons are removed, an ion is formed that is smaller than the source element.The radius of an atom in a crystal lattice is measured to calculate its ionic radius.This only occurs if the elements are of the same ion type, i.e., cations or anions. Like other forms of atomic radii, Ionic radii grow as you descend a group and drop as you go across. Similarly, when an electron is added to an atom to form an anion, the additional electron resists other electrons, causing the atom to grow in size. When an atom loses electrons to create a cation, the lost electron no longer shields the other electrons from the nucleus' charge as a result, the other electrons are more attracted to the nucleus, and the atom's radius shrinks. Since there are more electrons, there are more electron-electron repulsions, which causes the electron cloud to expand out. When the electrons exceed the protons, the protons' total attractive pull for the electrons decreases. The protons now dominate electrons, and the remaining electrons are drawn closer to the nucleus. When the valence electron(s) are eliminated, and the resultant ion has one less occupied primary energy level, resulting in a smaller electron cloud. Picometers (pm) or Angstroms (Å) are the most common units to express ionic radii. The spacing between ions in a crystal lattice can be calculated by adding the ionic radii of a cation and an anion. Although atoms and ions do not have specific borders, it is beneficial to treat them as spherical particles with radii. The radius of an ion, whether anion or cation, is called the ionic radius (rion). The size of any molecule is influenced by the strength of the attraction and the relative numbers of the two particles (protons and electrons) in a specific atom or ion. Protons and electrons have a high affinity to one another. In this article, we will discuss more about atomic radii vs ionic radii. Ions and atoms, too, have radii, and many of their characteristics are based on this. A tennis ball certainly cannot be used to play soccer. Their size restricts their application to a specific sport. Even though they are balls, they come in a variety of shapes, sizes, and applications. Basketballs, tennis balls, handballs, soccer balls, footballs, and a variety of others are among the most well-known. Use Internuclear distance formula to find the r +.įigure 3: The Hard sphere model can roughly determine the ion radii.Throughout the history of sports, various types of sports balls have been used for various games.Find internuclear distance (d) between anion and cation.This will minize the distribution of electrons. *To calculate ion radii, Lande used ionic compound under solid state (ex: NaCl). Internuclear distance (d) = r cation + r anion 2 In general, scientists uses formula of Internuclear distance to test out the radii of ion then compared with the ion radii had done on X-ray diffraction: Figure 2: 3-D hard sphere model of Sodium Chloride, taken with permission from en./wiki/File:So.e-3D-ionic.png The Hard-Sphere model can be applied to metallic and ionic compounds such as NaCl, which is shown below. Figure 1: Schematic of the hard-sphere model 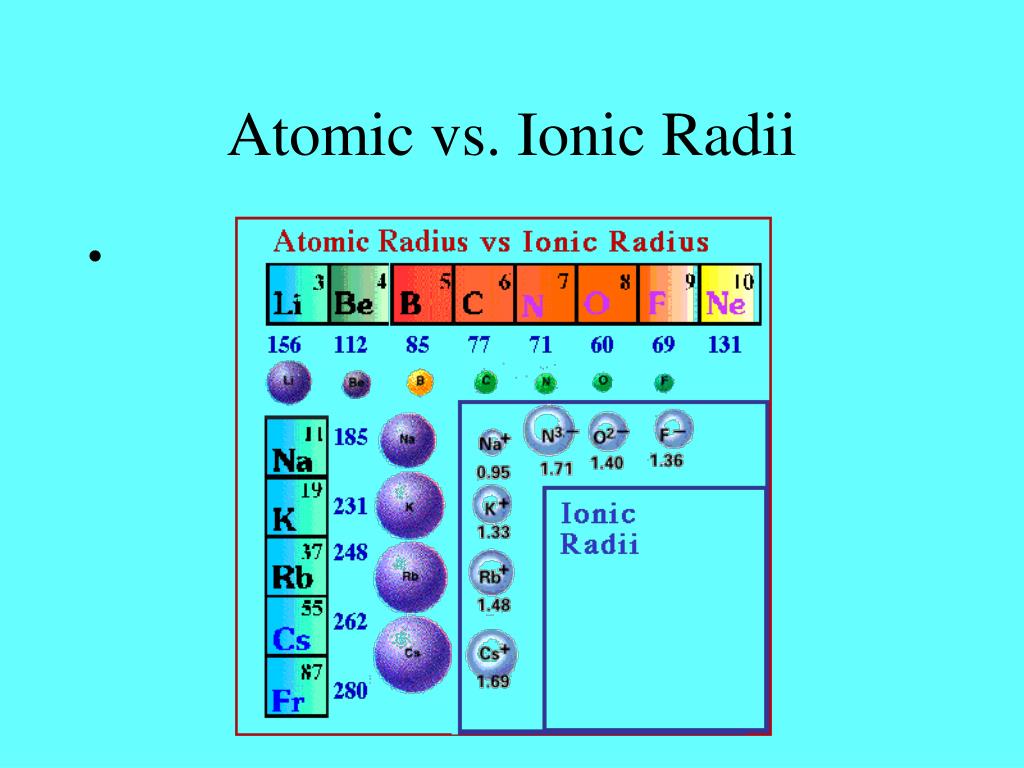
The ion radii measured under crystal state of ionic compound which cations and anions are stacking in pattern as shown below. 5 The Hard-Sphere model has been tested by well-known scientists Lande', Pauling and Goldsmidt. The Hard-Sphere model are impenetrable spheres that do not overlap in space. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
